Example with custom stratification by isolate source
Source:vignettes/SalmonellaExamples.Rmd
SalmonellaExamples.RmdAnalysing a small user-generated geno-pheno dataset of Salmonella enterica
Introduction
This vignette shows how AMRgen can be used to explore a
small user-generated dataset with genotypic and phenotypic data for
non-typhoidal, non-invasive Salmonella enterica isolates.
Phenotypic data consists of MIC E-test results for ciprofloxacin,
levofloxacin, and moxifloxacin. Genotypic data includes AMRFinderPlus
results for quinolone resistance markers.
Start by loading the package:
Import data
In this example, the data table was exported from the user’s
laboratory information management system as a single .csv
file that includes both genotypic and phenotypic results for 115 S.
enterica isolates, with one line per isolate. There are columns for
two categorical variables of interest: isolate source (human, animal, or
other) and isolate serovar. Genotype data is stored in a single column,
and where multiple quinolone resistance markers were identified, these
are listed in a string with semicolon delimiters. Phenotype data is
stored in separate columns for each antimicrobial agent that was tested
(ciprofloxacin, levofloxacin, moxifloxacin).
Data in this format can be read into R using read_csv()
and then formatted for use with AMRgen. Here, the object
salm_raw is already pre-loaded with the package so there is
no need to read in the .csv file.
# Importing raw data from .csv file (not needed to run vignette)
salm_raw <- read_csv("Salmonella_pheno_geno_data.csv")
head(salm_raw)
#> # A tibble: 6 × 7
#> Sample Source Serovar CpL_Genotype Ciprofloxacin Levofloxacin Moxifloxacin
#> <chr> <chr> <chr> <chr> <chr> <chr> <chr>
#> 1 SAL001 Animal other gyrA_S83F;p… 0.19 0.5 0.5
#> 2 SAL002 Human S. Enterit… gyrA_S83Y 0.38 1.5 1.5
#> 3 SAL003 Human other gyrA_S83F;p… 3 12 24
#> 4 SAL004 other S. Infantis gyrA_D87G 0.125 0.5 0.5
#> 5 SAL005 Human other gyrA_D87Y 0.094 0.25 0.38
#> 6 SAL006 Human S. Kentucky gyrA_S83F;g… 8 8 32The AMRgen package requires separate phenotypic and
genotypic tables with specific column headers, so the next step is to
separate and wrangle these data into the required formats.
Genotype table
Format raw genotype data for AMRgen
To use downstream AMRgen functions, a genotype table
must have at a minimum the following columns: Name (unique
sample name for each isolate), marker (name of the
resistance marker detected), and drug_class (antibiotic
class associated with this marker). It also needs to be in long form,
with a row for each isolate/marker combination. In our dataset, the
genotype information is found in a single column alongside the metadata
and phenotypic information, and we can have multiple markers per row (up
to four). Here, we carry out the following steps to generate a genotype
table that is compatible with AMRgen functions:
- Select only the genotype column (called
CpL_Genotypein this dataset) and the column specifying the individual isolate names (calledSamplein this dataset) - Rename the column
SampletoName, which is the default column heading used byAMRgenfor identifying individual isolates - Separate the genotype column
CpL_Genotypeinto individual columns for each marker (up to four per isolate in these data), specifying the delimiter; - Convert this wide-form data (one row per isolate) to a long-form
table (one row per isolate/marker combination) using
pivot_longer - Add a column specifying that all these markers are associated with resistance to quinolones
# Extract geno data, separate by delimiter, pivot longer, and add drug_class column
salm_geno <- salm_raw %>%
select(Sample, CpL_Genotype) %>%
rename(Name = Sample) %>%
separate_wider_delim(CpL_Genotype,
delim = ";", names = c("Marker_1", "Marker_2", "Marker_3", "Marker_4"),
too_few = "align_start", cols_remove = TRUE
) %>%
pivot_longer(
cols = c(Marker_1, Marker_2, Marker_3, Marker_4),
names_to = "marker_no",
values_to = "marker",
values_drop_na = TRUE
) %>%
select(-marker_no) %>%
mutate(
drug_class = "Quinolones",
drug = NA_character_
) ## the variable drug appears essential for get_binary_matrix function to work...
# Check the format of the processed genotype table
head(salm_geno)
#> # A tibble: 6 × 4
#> Name marker drug_class drug
#> <chr> <chr> <chr> <chr>
#> 1 SAL001 gyrA_S83F Quinolones NA
#> 2 SAL001 parC_T57S Quinolones NA
#> 3 SAL002 gyrA_S83Y Quinolones NA
#> 4 SAL003 gyrA_S83F Quinolones NA
#> 5 SAL003 parC_T57S Quinolones NA
#> 6 SAL003 qnrB19 Quinolones NASummarise genotype data
The summarise_geno() function can provide various
summaries of the genotype table, including the total number of unique
markers and a table showing each marker’s prevalence in the dataset.
# Create geno_summary object
salm_geno_summary <- summarise_geno(salm_geno)
# Total number of markers, drugs, and drug classes
salm_geno_summary$uniques
#> # A tibble: 3 × 2
#> column n_unique
#> <chr> <int>
#> 1 marker 26
#> 2 drug 1
#> 3 drug_class 1
# Prevalence of each detected marker, in decreasing abundance
salm_geno_summary$markers %>% arrange(-n)
#> # A tibble: 26 × 4
#> marker drug drug_class n
#> <chr> <chr> <chr> <int>
#> 1 parC_T57S NA Quinolones 49
#> 2 gyrA_S83F NA Quinolones 18
#> 3 gyrA_S83Y NA Quinolones 18
#> 4 qnrS1 NA Quinolones 18
#> 5 qnrB19 NA Quinolones 16
#> 6 gyrA_D87Y NA Quinolones 14
#> 7 gyrA_D87N NA Quinolones 12
#> 8 gyrA_D87G NA Quinolones 11
#> 9 parC_S80I NA Quinolones 9
#> 10 qnrA1 NA Quinolones 5
#> # ℹ 16 more rowsThese summaries show that there are 26 distinct genotypic markers for quinolone resistance in this dataset. The most common one is parC_T57S, found in 49 of 115 isolates, followed by gyrA_S83F, gyrA_S83Y, and qnrS1, each found in 18 of 115 isolates. Eleven markers are found only once across all isolates.
Phenotype table
Format raw phenotype data for AMRgen
To use downstream AMRgen functions, a phenotype table
must be in long form with a single column for all antimicrobial agents
that were tested, rather than separate columns for each agent. It also
needs to have at a minimum the following columns: id
(unique sample name for each isolate), spp_pheno (species
name formatted as per the AMR package mo class),
drug (antimicrobial agent name formatted as per the AMR
package ab class), a S/I/R phenotype column (e.g. one or
more of pheno_eucast, pheno_clsi,
pheno_provided, ecoff). If raw assay data is
to be included, it needs to be in a column called mic
and/or disk.
To convert out dataset to this format, we begin by extracting the
columns with the unique isolate names (Sample) and those
with the MIC results for the three antimicrobial agents that we tested
(Ciprofloxacin, Levofloxacin,
Moxifloxacin). We also keep the columns with additional
metadata (Source, Serovar), as these are not
in the genotype table. The antimicrobial columns need to be forced to
character vectors to avoid issues caused by occasional presence of
non-numeric prefixes (> or <). We then
pivot the table to long form.
# Pheno table: select columns with sample ID, metadata, and antimicrobials tested,
# then pivot to long form
salm_pheno <- salm_raw %>%
select(Sample, Source, Serovar, Ciprofloxacin, Levofloxacin, Moxifloxacin) %>%
mutate(across(c(Ciprofloxacin, Levofloxacin, Moxifloxacin), as.character)) %>%
pivot_longer(
cols = c(Ciprofloxacin, Levofloxacin, Moxifloxacin),
names_to = "drug",
values_to = "MIC.values",
values_drop_na = TRUE
)Our phenotype data is not yet in a standard AMR/AMRgen format so we
use the helpful format_pheno function to add the species
name, to format the antimicrobial and MIC columns correctly, and to
generate the S/I/R phenotype column pheno_eucast by
interpreting our Salmonella MIC data against EUCAST breakpoints. We
apply these breakpoints across our human/animal/other isolates, as our
interest is in resistance phenotypes that are potentially problematic in
human infections, including zoonotic ones.
salm_ast <- format_pheno(
input = salm_pheno,
sample_col = "Sample",
species = "Salmonella enterica",
ab_col = "drug",
mic_col = "MIC.values",
interpret_eucast = TRUE
)
# Check the format of the processed phenotype table
head(salm_ast)
#> # A tibble: 6 × 7
#> id drug mic pheno_eucast spp_pheno Source Serovar
#> <chr> <ab> <mic> <sir> <mo> <chr> <chr>
#> 1 SAL001 CIP 0.19 R B_SLMNL_ENTR Animal other
#> 2 SAL001 LVX 0.50 S B_SLMNL_ENTR Animal other
#> 3 SAL001 MFX 0.50 R B_SLMNL_ENTR Animal other
#> 4 SAL002 CIP 0.38 R B_SLMNL_ENTR Human S. Enteritidis
#> 5 SAL002 LVX 1.50 R B_SLMNL_ENTR Human S. Enteritidis
#> 6 SAL002 MFX 1.50 R B_SLMNL_ENTR Human S. EnteritidisSummarise phenotype data
The summarise_pheno function can be used to generate
various summaries of the phenotype table, including the total number of
samples, drugs, and species, and table of S/I/R counts for each drug in
the dataset. Our dataset is limited to a single species with data from
only one method (E-test MIC), and we are looking only at the EUCAST
breakpoints, but the vignette “Analysing Geno-Pheno Data” shows how the
summarise_pheno function can be used for datasets with
multiple species, methods, drug classes, and interpretation
guidelines.
salm_pheno_summary <- summarise_pheno(salm_ast, pheno_cols = c("pheno_eucast"))
#> No disk data colummn provided
# Number of samples, drugs, species, and methods included in phenotype table
salm_pheno_summary$uniques
#> # A tibble: 3 × 2
#> column n_unique
#> <chr> <int>
#> 1 id 115
#> 2 drug 3
#> 3 spp_pheno 1
# SIR summary table for each drug in the phenotype table
salm_pheno_summary$pheno_counts_list
#> $pheno_eucast
#> # A tibble: 3 × 6
#> drug drug_name spp_pheno S R I
#> <ab> <chr> <chr> <int> <int> <int>
#> 1 CIP Ciprofloxacin Salmonella enterica 23 92 NA
#> 2 LVX Levofloxacin Salmonella enterica 63 25 27
#> 3 MFX Moxifloxacin Salmonella enterica 20 95 NAThese summaries show that there are 115 isolates of one species in this dataset, with results for three drugs. Similar proportions were resistant to ciprofloxacin and moxifloxacin, whereas a lower proportion was resistant to levofloxacin (using the EUCAST breakpoints).
Now that we have formatted our data into a genotype table
(salm_geno) and a phenotype table (salm_ast)
that are compatible with AMRgen, we can use its downstream
functions for plotting distributions, combining geno/pheno data,
modeling binary drug phenotype, assessing positive predictive values of
genetic markers, and comparing how combinations of markers influence
resistance phenotype. Other vignettes outline these workflows in more
detail, so here we show only a basic workflow, focusing on how
additional metadata can be included.
Plot phenotype data distributions
Overall phenotype distributions
We begin by looking at the distribution of the MIC values in our
dataset for the three antimicrobials that we tested. If desired, these
plots could then be combined into a multipanel figure using packages
like patchwork or ggpubr.
# Plot MIC distributions coloured by S/I/R call
assay_by_var(pheno_table = salm_ast, pheno_drug = "Ciprofloxacin", measure = "mic", colour_by = "pheno_eucast")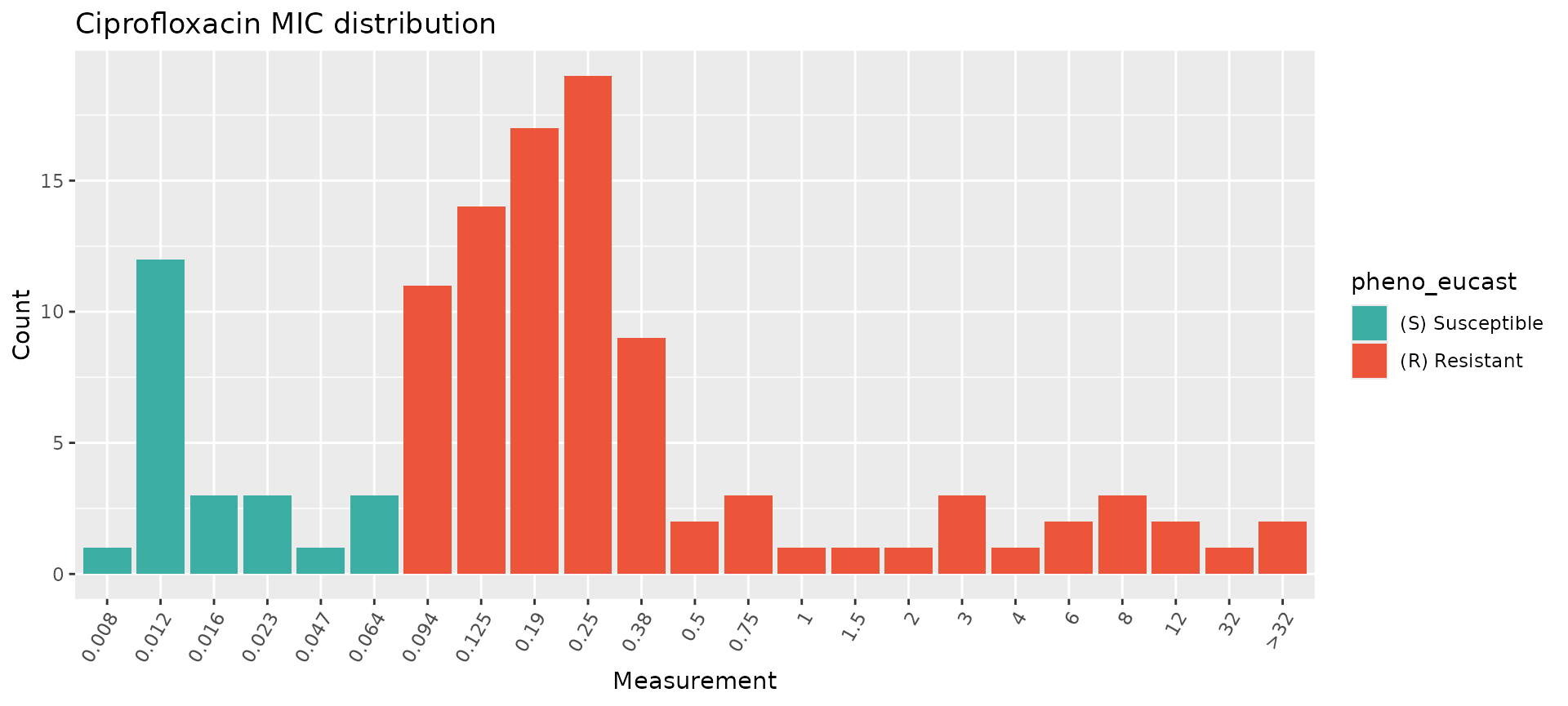
assay_by_var(pheno_table = salm_ast, pheno_drug = "Levofloxacin", measure = "mic", colour_by = "pheno_eucast")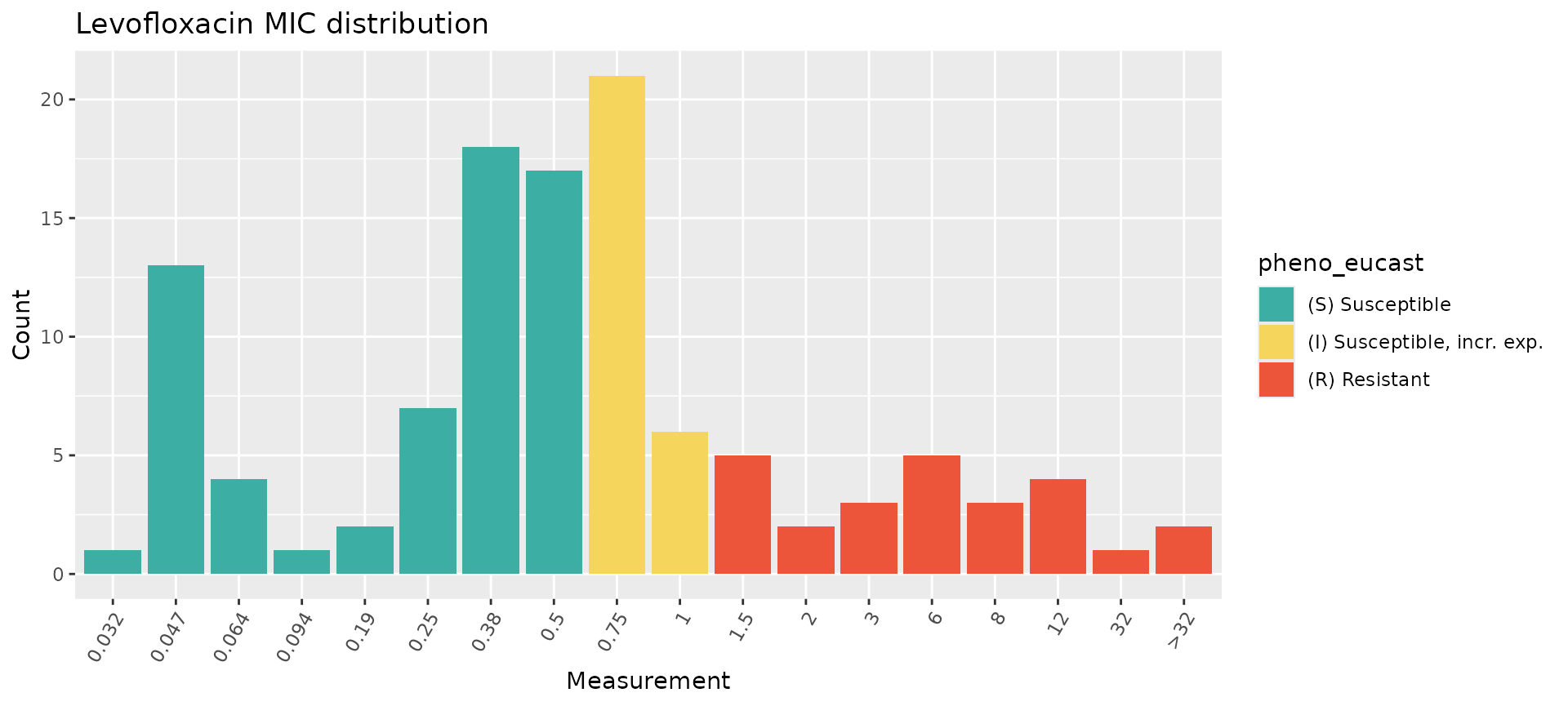
assay_by_var(pheno_table = salm_ast, pheno_drug = "Moxifloxacin", measure = "mic", colour_by = "pheno_eucast")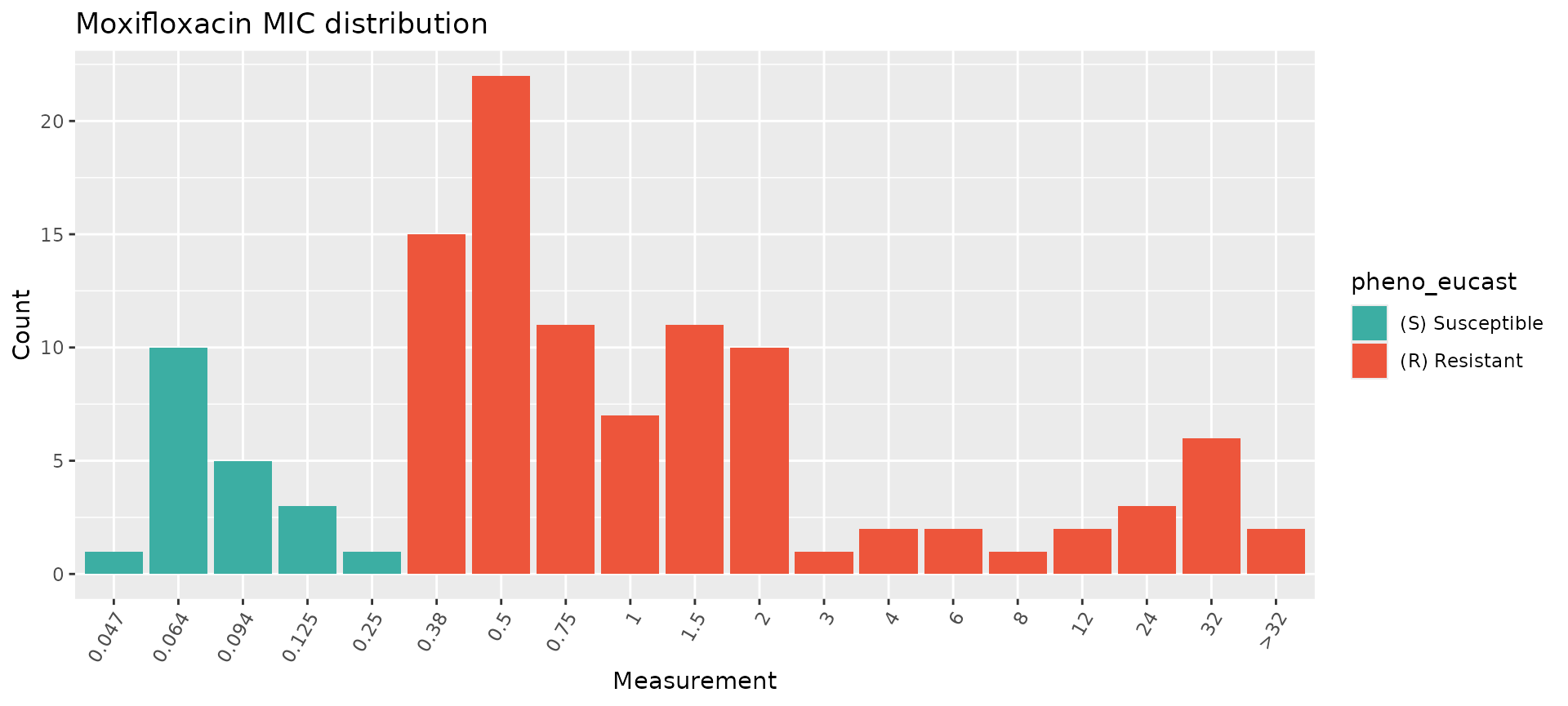
While these plots show the MIC distributions of all isolates in our dataset, we can break this down further by isolation source (human, animal, other) and by S. enterica serovar. This can be done in a few different ways, notably by faceting and/or by modifying the colour variable.
Phenotype distributions by categorical variable
The assay_by_var function can separate plots by an
additional categorical variable by specifying facet_by
within the function. For example, we can split the ciprofloxacin plot by
isolation source like this:
assay_by_var(pheno_table = salm_ast, pheno_drug = "Ciprofloxacin", measure = "mic", colour_by = "pheno_eucast", facet_by = "Source")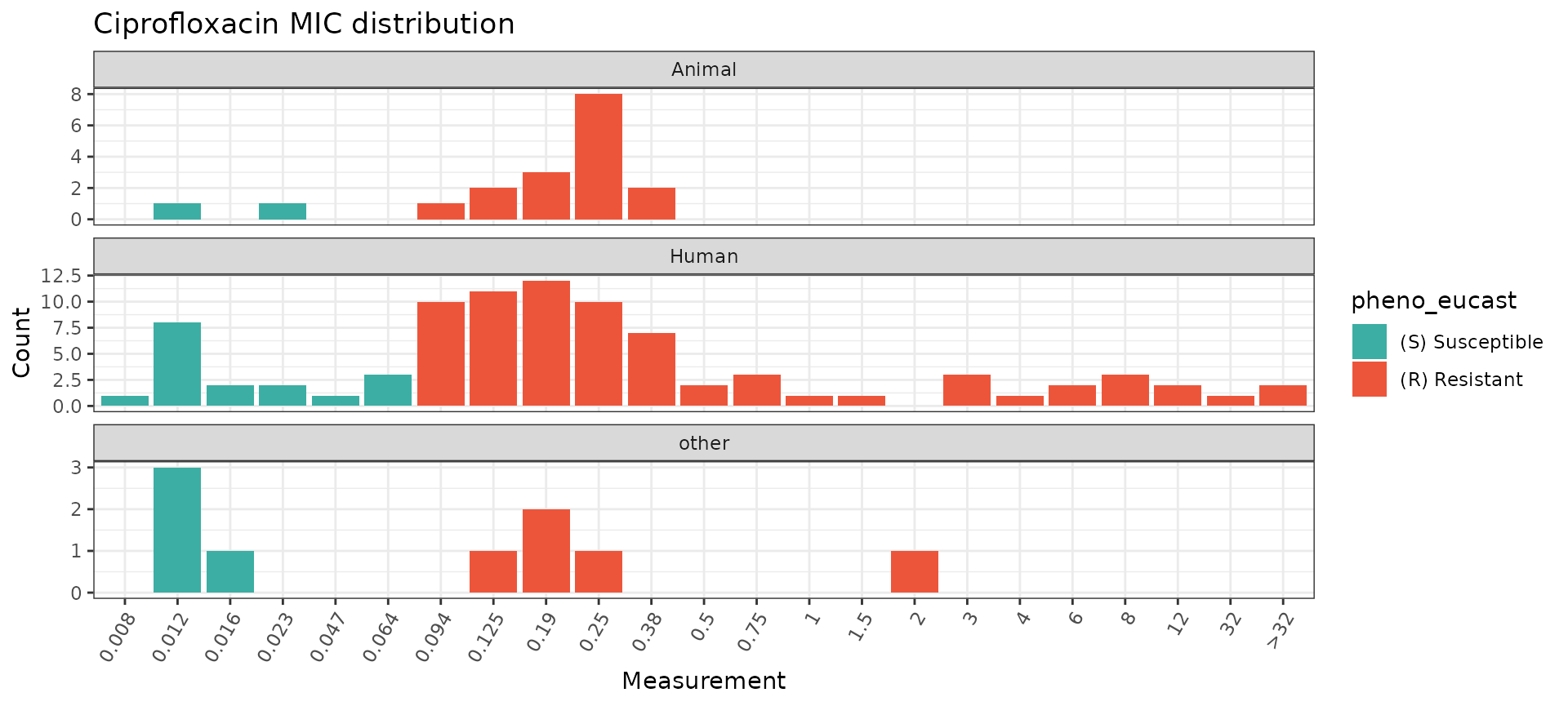
Alternatively, because assay_by_var is a
ggplot2 function, it can be extended by adding
ggplot2 layers, including facet_wrap() for a
single faceting variable (to allow more control over how the facets are
displayed) or facet_grid() to facet on two variables.
assay_by_var(pheno_table = salm_ast, pheno_drug = "Ciprofloxacin", measure = "mic", colour_by = "pheno_eucast") +
facet_grid(Source ~ Serovar)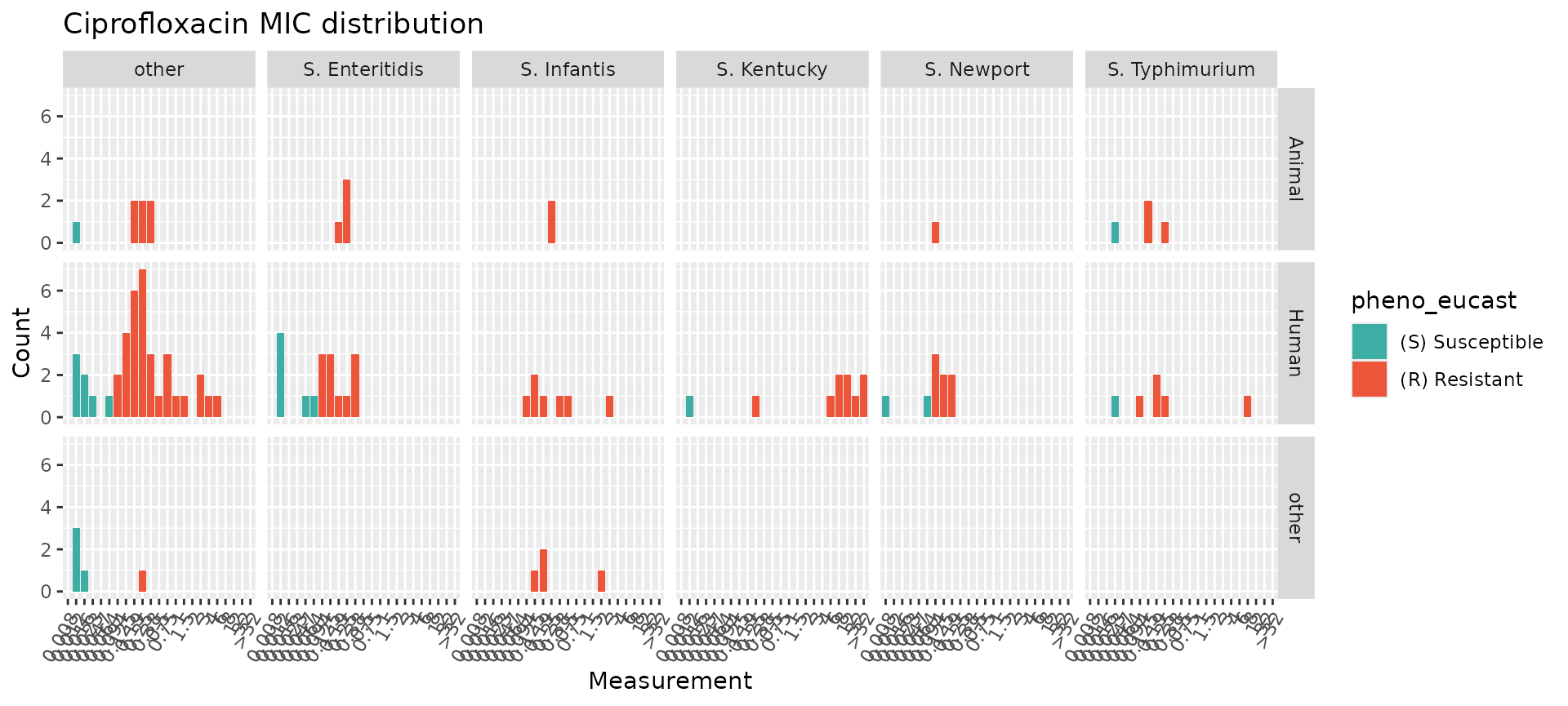
Another option for displaying categorical metadata is to change the
colour variable colour_by but still show the breakpoints as
vertical lines using the guideline and species
calls within assay_by_var. If desired, you can also modify
other aspects of the plot using standard ggplot2 extensions (e.g. using
viridis to change the colour palette).
# Specify species and guideline to show breakpoints, but colour bars by isolation source
assay_by_var(pheno_table = salm_ast, pheno_drug = "Ciprofloxacin", measure = "mic", colour_by = "Source", species = "Salmonella enterica", guideline = "EUCAST 2025") +
scale_fill_viridis_d(end = 0.8)
#> MIC breakpoints determined using AMR package: S <= 0.064 and R > 0.064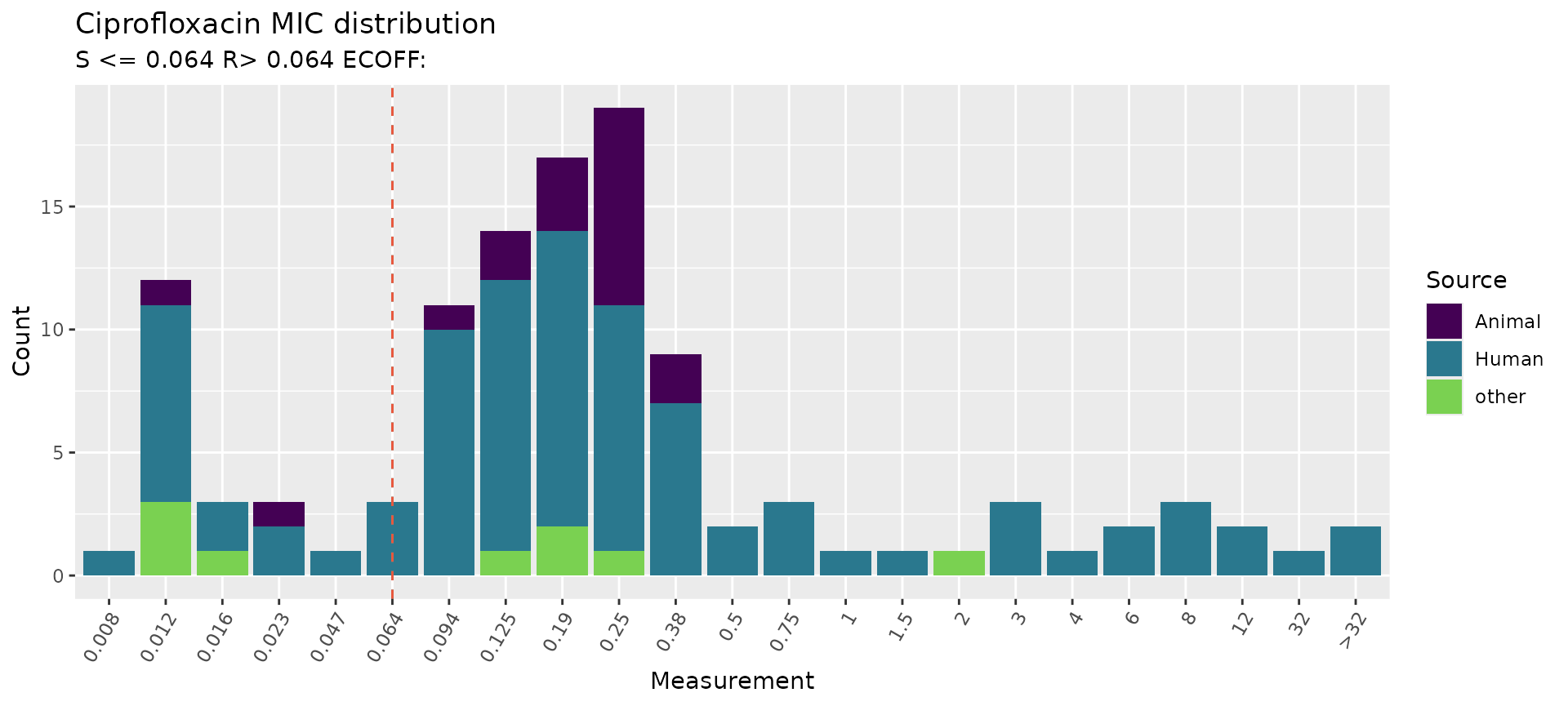
Combine ciprofloxacin genotype and phenotype data
Analysis of combined genotype-phenotype data must be carried out
separately for each antimicrobial agent. The first step is to generate a
combined dataframe for the specified agent from the genotype and
phenotype tables, using the get_binary_matrix() function.
As an example, we will do this for ciprofloxacin with our small S.
enterica dataset, but this could also be done for levofloxacin and
moxifloxacin.
### get_binary_matrix throws an error unless the variable drug_class is in salm_geno, even if it's empty
cip_bin <- get_binary_matrix(
salm_geno,
salm_ast,
pheno_drug = "Ciprofloxacin",
geno_class = "Quinolones",
sir_col = "pheno_eucast",
keep_assay_values = TRUE,
keep_assay_values_from = "mic"
)
#> Defining NWT in binary matrix as I/R vs S, as no ECOFF column defined
# check format
head(cip_bin)
#> # A tibble: 6 × 31
#> id pheno mic R NWT gyrA_S83F parC_T57S gyrA_S83Y qnrB19 gyrA_D87G
#> <chr> <sir> <mic> <dbl> <dbl> <dbl> <dbl> <dbl> <dbl> <dbl>
#> 1 SAL001 R 0.190 1 1 1 1 0 0 0
#> 2 SAL002 R 0.380 1 1 0 0 1 0 0
#> 3 SAL003 R 3.000 1 1 1 1 0 1 0
#> 4 SAL004 R 0.125 1 1 0 0 0 0 1
#> 5 SAL005 R 0.094 1 1 0 0 0 0 0
#> 6 SAL006 R 8.000 1 1 1 0 0 0 0
#> # ℹ 21 more variables: gyrA_D87Y <dbl>, gyrA_D87N <dbl>, qnrS1 <dbl>,
#> # qnrA1 <dbl>, gyrA_D87X <dbl>, qepA1 <dbl>, parC_S80I <dbl>, gyrA_u <dbl>,
#> # gyrA_D82N <dbl>, qnrB2 <dbl>, qnrD <dbl>, gyrB_E446D <dbl>, qnrDv1 <dbl>,
#> # parE_S474G <dbl>, gyrA_Y100H <dbl>, parC_S80X <dbl>, parC_F60L <dbl>,
#> # parE_D475Y <dbl>, qnrS4 <dbl>, gyrB_L446M <dbl>, parC_A81X <dbl>
# list colnames (alphabetically) to see full list of quinolone markers in data
# (will include additional columns id, pheno, mic, NWT, R, Source, and Serovar)
sort(colnames(cip_bin))
#> [1] "gyrA_D82N" "gyrA_D87G" "gyrA_D87N" "gyrA_D87X" "gyrA_D87Y"
#> [6] "gyrA_S83F" "gyrA_S83Y" "gyrA_u" "gyrA_Y100H" "gyrB_E446D"
#> [11] "gyrB_L446M" "id" "mic" "NWT" "parC_A81X"
#> [16] "parC_F60L" "parC_S80I" "parC_S80X" "parC_T57S" "parE_D475Y"
#> [21] "parE_S474G" "pheno" "qepA1" "qnrA1" "qnrB19"
#> [26] "qnrB2" "qnrD" "qnrDv1" "qnrS1" "qnrS4"
#> [31] "R"This binary matrix can be used for numerous downstream analyses, as described in other vignettes. Here, we show how some can be modified with additional metadata variables.
The get_binary_matrix() output (cip_bin)
did not retain our additional metadata variables Source and
Serovar. To use these variables in downstream analyses, we
first extract them from salm_pheno and then join to
cip_bin to create cip_bin_meta, which can be
used in plotting functions (warning: statistical model functions may not
work correctly with these additional metadata so cip_bin
should be used in those instead of cip_bin_meta).
salm_metadata <- salm_pheno %>%
select(Sample, Source, Serovar) %>%
rename(id = Sample)
cip_bin_meta <- left_join(cip_bin, salm_metadata)
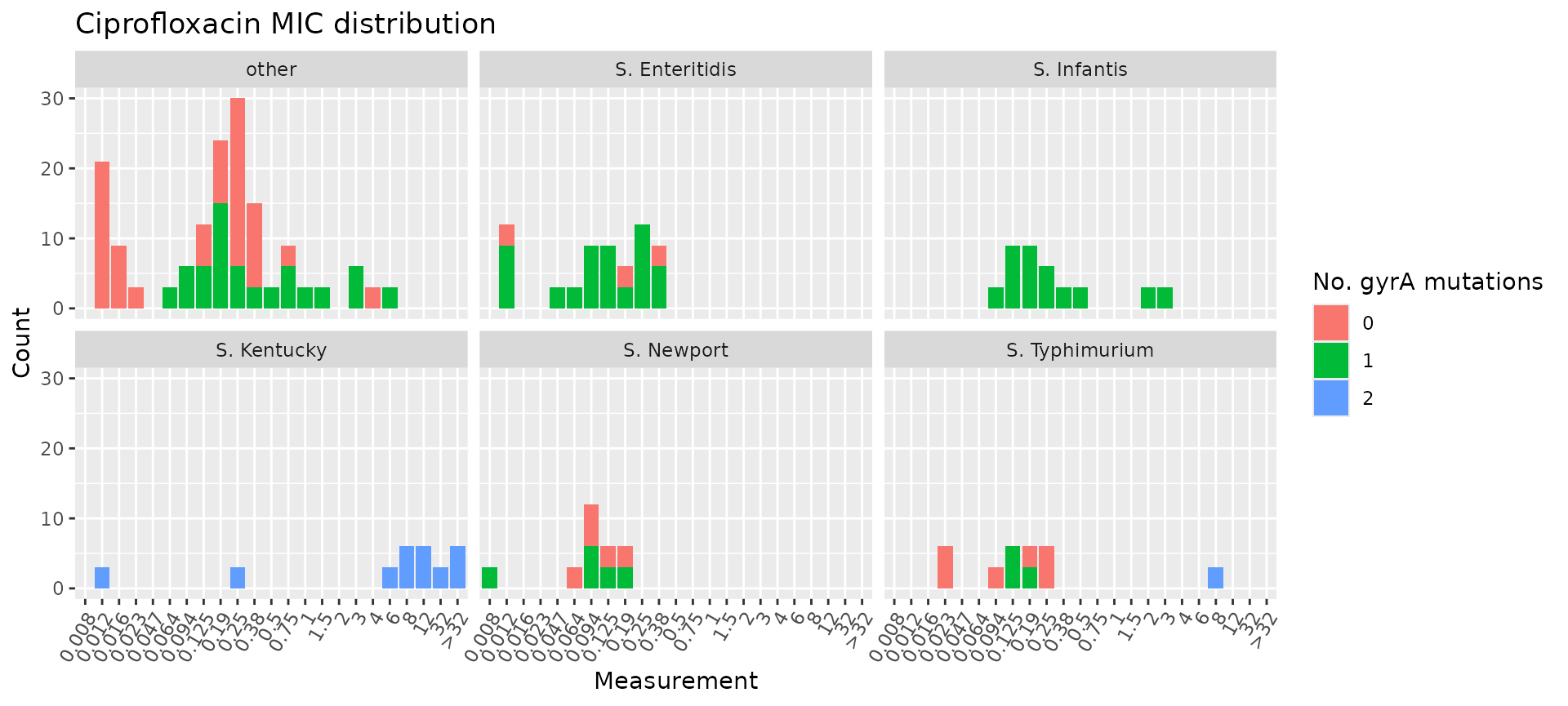
#> Joining with `by = join_by(id)`Plot ciprofloxacin phenotype by number of mutations and Serovar/Source
Joining the metadata to cip_bin allows us to colour the
MIC distribution plot by the number of mutations in gyrA and
facet by Serovar, highlighting that all S. Infantis
isolates had one gyrA mutation and all S. Kentucky isolates had
two gyrA mutations, whereas other serovars had variable numbers
of mutations.
# count the number of gyrA mutations per genome
gyrA_mut <- cip_bin_meta %>%
dplyr::mutate(gyrA_mut = rowSums(across(contains("gyrA_") & where(is.numeric)), na.rm = T)) %>%
select(mic, gyrA_mut, Source, Serovar)
# plot the MIC distribution, coloured by count of gyrA mutations
mic_by_gyrA_count <- assay_by_var(gyrA_mut, measure = "mic", colour_by = "gyrA_mut", colour_legend_label = "Number of\ngyrA mutations", measure_axis_label = "MIC (mg/L)", pheno_drug = "Ciprofloxacin", colours = viridisLite::viridis(5)[c(4, 3, 2)]) + facet_wrap(~Serovar)
# add title with italicised species and drug names
mic_by_gyrA_count + ggtitle(expression(paste(
"Ciprofloxacin MIC in ",
italic("Salmonella"),
" serovars, by number of ",
italic("gyrA"),
" mutations"
)))
Similarly, we can plot the total number of markers per isolate and
facet by Source.
# count the number of genetic determinants per genome
marker_count <- cip_bin_meta %>%
mutate(marker_count = rowSums(across(where(is.numeric) & !any_of(c("R", "NWT"))), na.rm = T)) %>%
select(mic, marker_count, Source, Serovar)
# plot the MIC distribution, coloured by count of associated genetic markers
mic_by_marker_count <- assay_by_var(marker_count, measure = "mic", colour_by = "marker_count", colour_legend_label = "Total number\nof markers", pheno_drug = "Ciprofloxacin", colours = viridisLite::viridis(max(marker_count$marker_count) + 1)) +
facet_wrap(~Source, ncol = 1)
mic_by_marker_count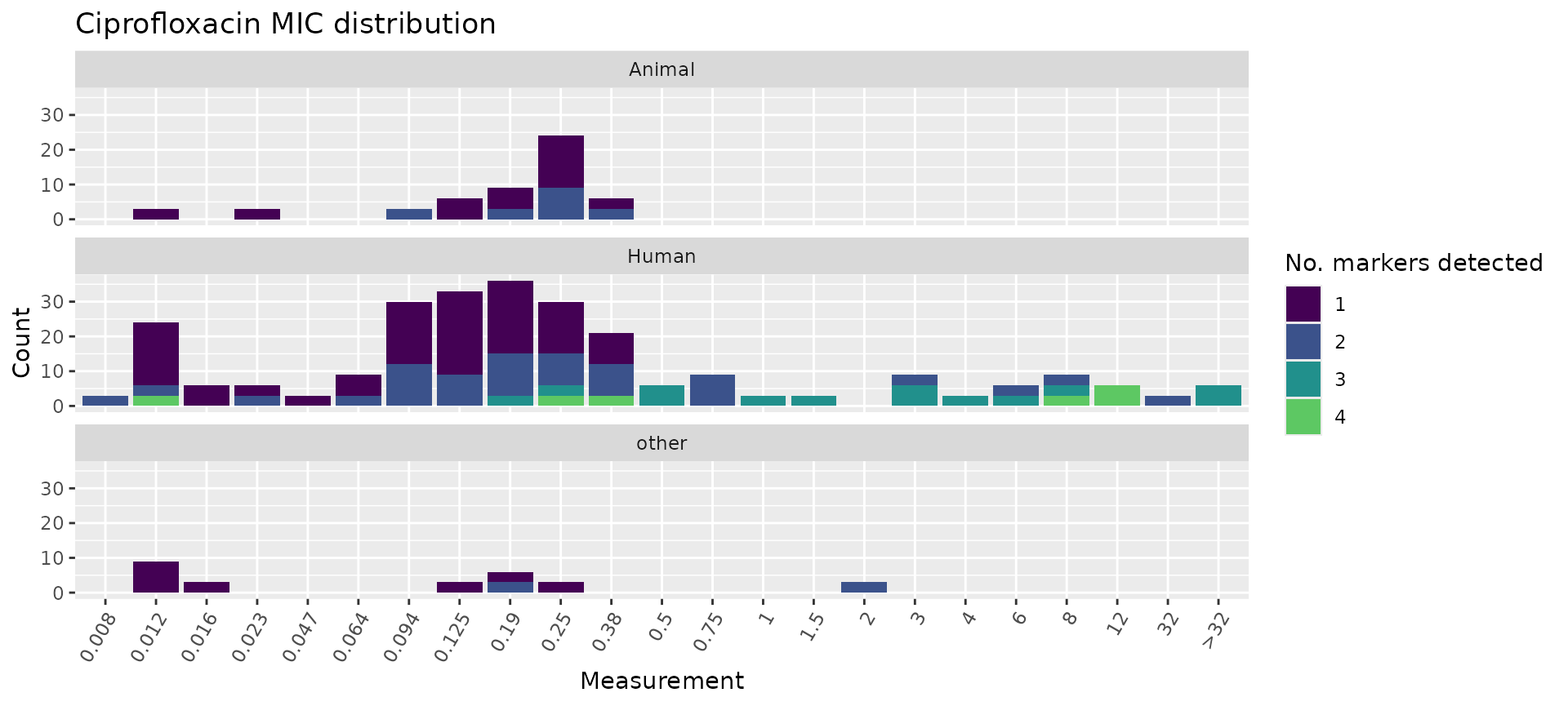
We can also use the boxplot=TRUE option in
assay_by_var() to see the MIC distribution as boxplots,
stratified by number of markers. This also summarises the median and
interquartile range of MIC values, per marker count so we can quantify
as well as visualise the impact of number of mutations on MIC.
# plot the MIC distributions as boxplots, stratified by number of markers
mic_boxplot_by_marker_count <- assay_by_var(marker_count, measure = "mic", colour_by = "marker_count", colour_legend_label = "Total number\nof markers", pheno_drug = "Ciprofloxacin", colours = viridisLite::viridis(max(marker_count$marker_count) + 1), boxplot = T)
mic_boxplot_by_marker_count$plot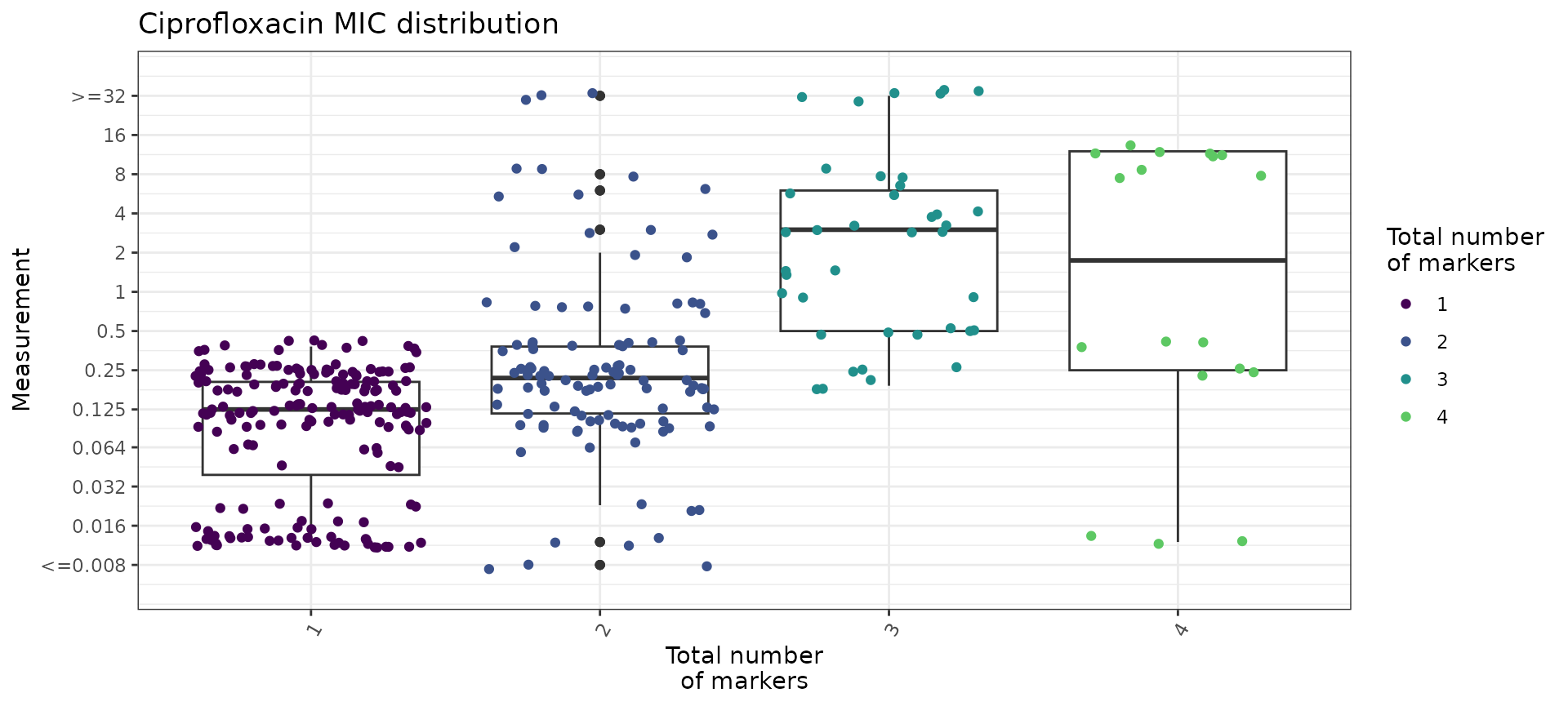
mic_boxplot_by_marker_count$stats
#> # A tibble: 4 × 6
#> marker_count n median geom_mean q25 q75
#> <dbl> <int> <dbl> <dbl> <dbl> <dbl>
#> 1 1 180 0.125 0.0884 0.041 0.205
#> 2 2 108 0.22 0.266 0.117 0.38
#> 3 3 39 3 2.22 0.5 6
#> 4 4 18 4.19 1.05 0.25 12We can also use the boxplot=TRUE option in
assay_by_var() to see the MIC distribution as boxplots,
stratified by number of markers. This also summarises the median and
interquartile range of MIC values, per marker count so we can quantify
as well as visualise the impact of number of mutations on MIC.
# plot the MIC distributions as boxplots, stratified by number of markers
mic_boxplot_by_marker_count_source <- assay_by_var(marker_count, measure = "mic", colour_by = "marker_count", colour_legend_label = "Total number\nof markers", pheno_drug = "Ciprofloxacin", colours = viridisLite::viridis(max(marker_count$marker_count) + 1), facet_by = "Source", boxplot = T)
mic_boxplot_by_marker_count_source$plot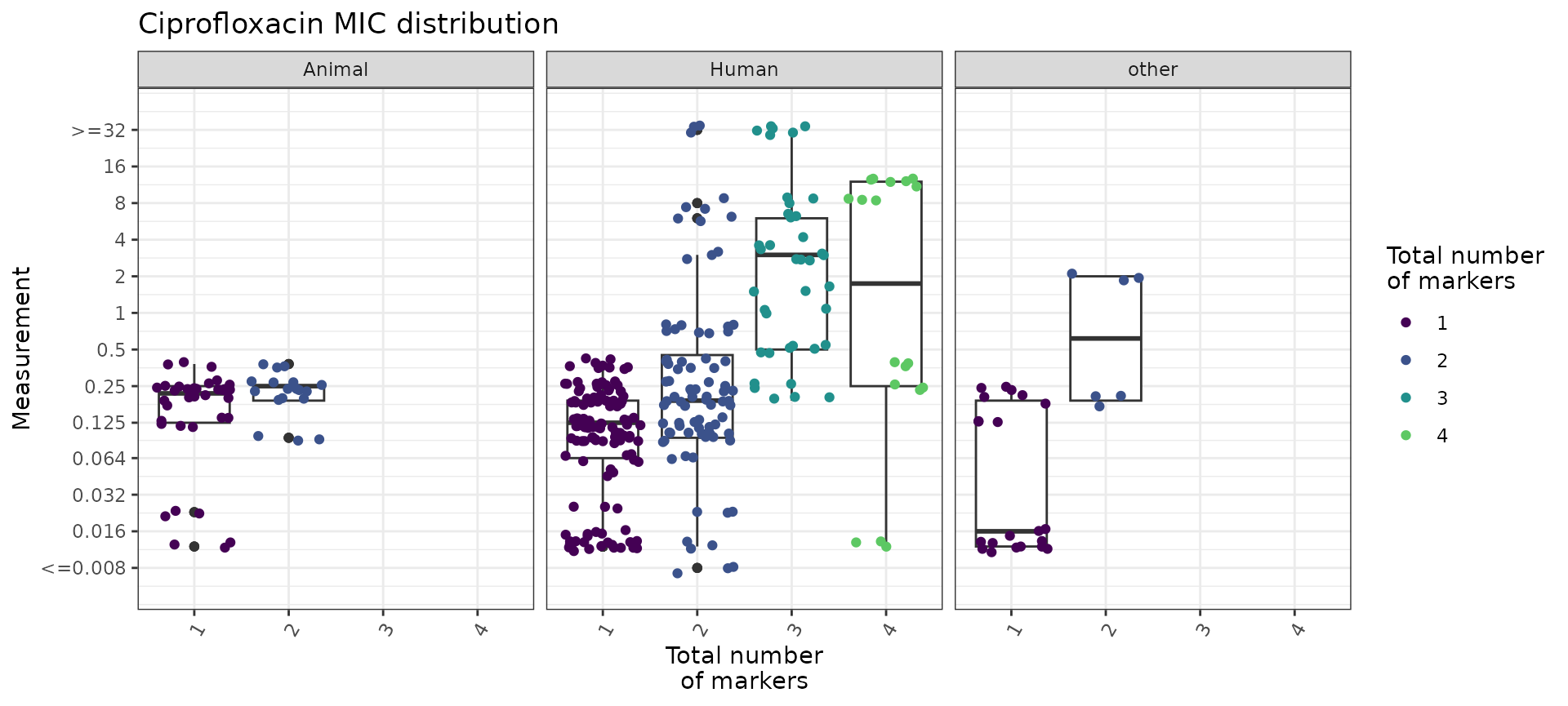
Plot ciprofloxacin phenotype by combinations of markers
We can look at how combinations of markers are associated with
phenotypic ciprofloxacin MIC by generating an UpSet plot with the
amr_upset function. This function does not accept extra
metadata columns so we use cip_bin instead of
cip_bin_meta here. As our dataset is quite small, we keep
all the combinations, including those with a single isolate
(min_set_size = 1).
# Compare ciprofloxacin MIC data with quinolone marker combinations,
# using the binary matrix we constructed earlier via get_binary_matrix()
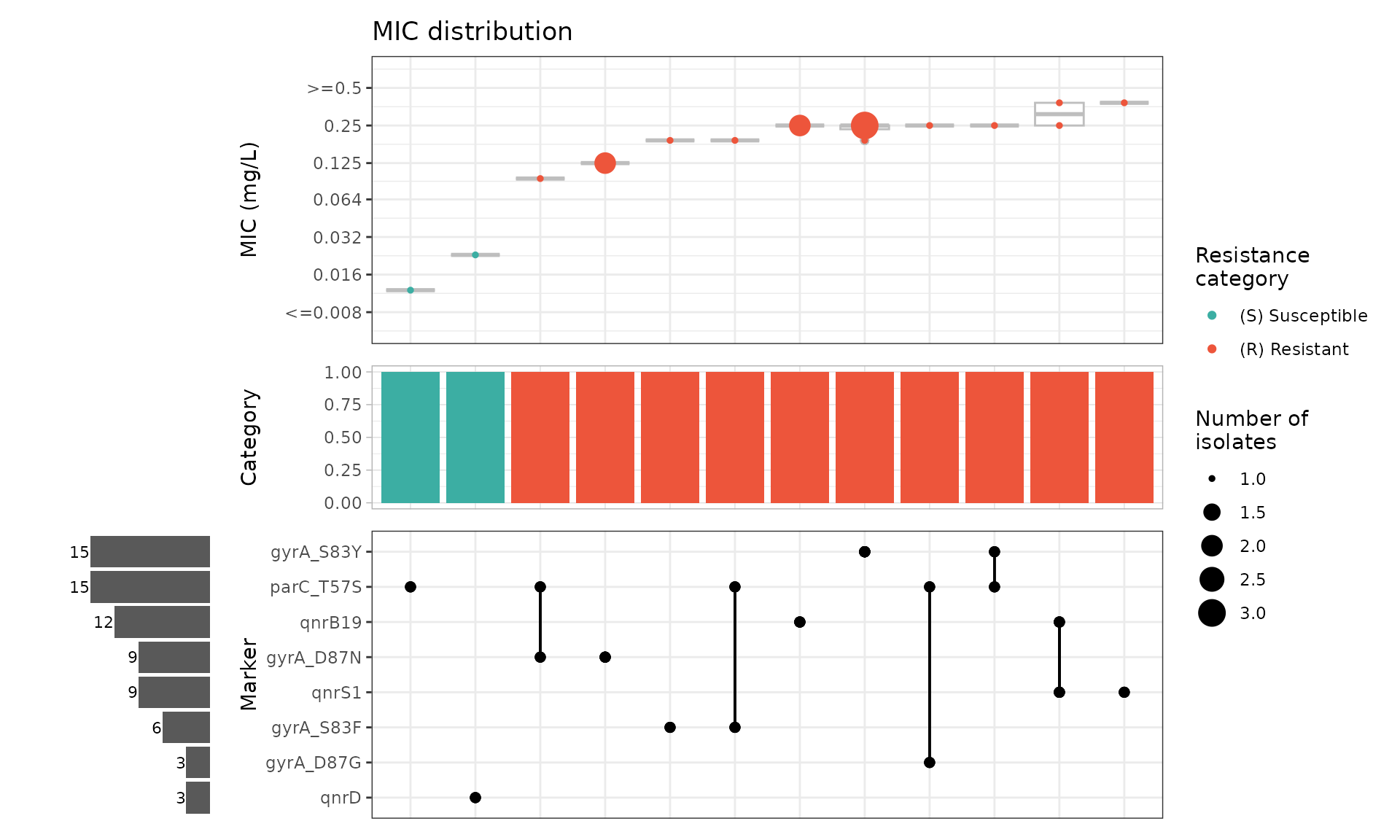
cipro_mic_upset <- amr_upset(
cip_bin,
min_set_size = 1,
assay = "mic",
order = "value"
)
#> Ordering markers by frequency
#> Scale for y is already present.
#> Adding another scale for y, which will replace the existing scale.
#> Scale for y is already present.
#> Adding another scale for y, which will replace the existing scale.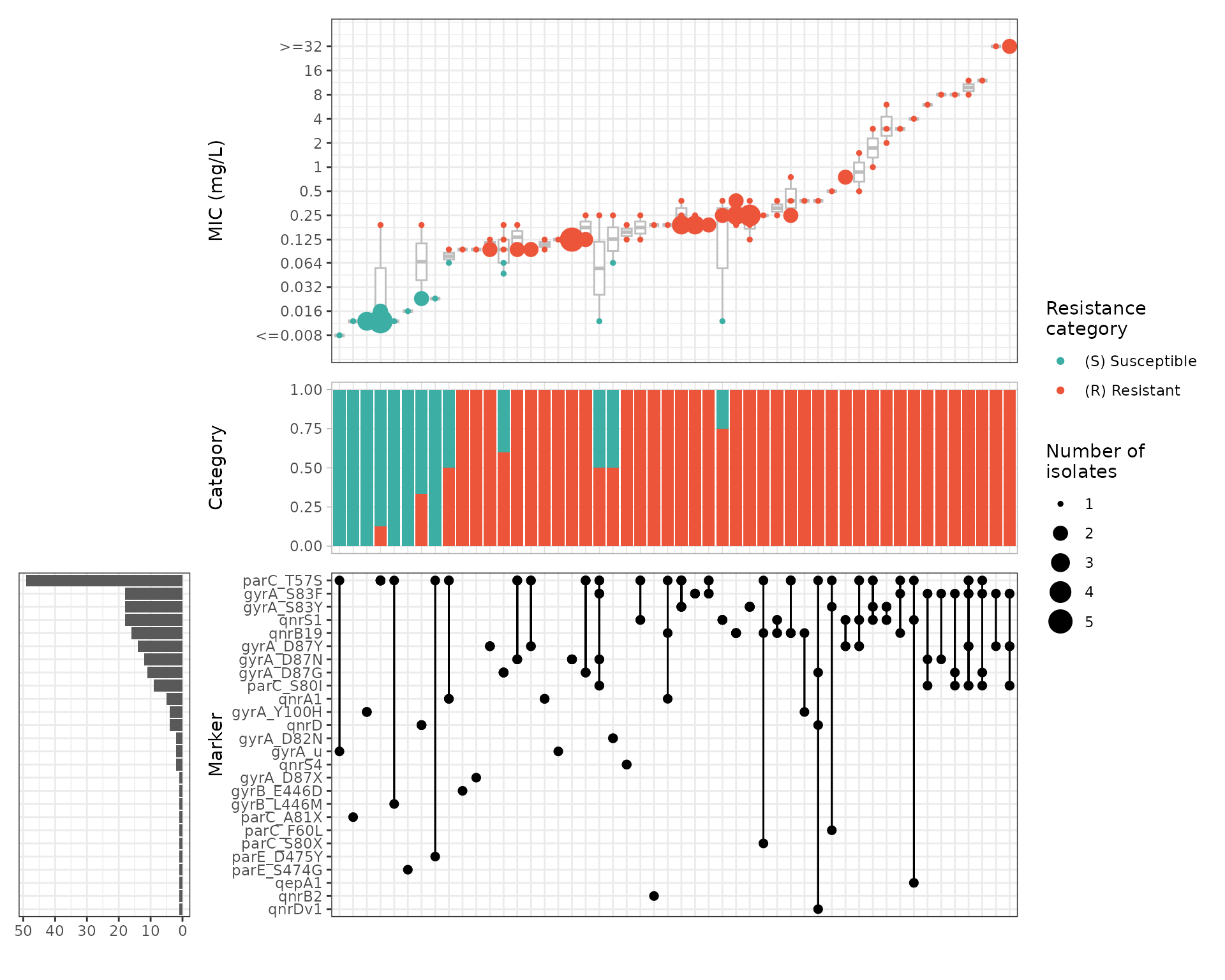
We can generate UpSet plots of a subset of isolates by filtering on
one of our additional metadata variables and then running
amr_upset(). For example, we can focus on the animal
isolates only:
cip_bin_animal <- cip_bin_meta %>%
filter(Source == "Animal") %>%
select(-Source, -Serovar)
cipro_mic_upset_animal <- amr_upset(
cip_bin_animal,
min_set_size = 1,
assay = "mic",
order = "value"
)
#> Ordering markers by frequency
#> Scale for y is already present.
#> Adding another scale for y, which will replace the existing scale.
#> Scale for y is already present.
#> Adding another scale for y, which will replace the existing scale.